How Many Protons In Nitrogen
Question: Nitrogen (Diminutive Number vii) has stable isotopes (half-life greater than 1 sec) of mass numbers 13, 14, 15, xvi and 17. Calculate number of protons and neutrons in each of the isotopes of nitrogen.
Solution:
The Atomic Number (Z) of Nitrogen is seven. The atomic number is e'er equal to the number of protons. And the number of protons always equals the electron number in an uncharged atom so that the cantlet is electrically neutral.
Therefore, an uncharged Nitrogen atom always has 7 protons and seven electrons.
An isotope is formed when the neutron number varies in an cantlet of the aforementioned element. This changes the mass number in an cantlet's nucleus since the mass number A is the sum of protons and neutrons. The proton number remains unchanged.
mass number A= Number of protons (p) + number of neutrons (n)
The mass number is written every bit a superscript next to the chemical element's symbol as,
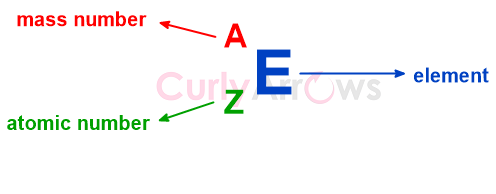
So, the number of neutrons in dissimilar Nitrogen isotopes xiii N, 14 N, xv Due north, 16 North, and 17 Northward is-
xiii N Isotope:
Mass number =xiii (Given)
mass number (A) = number of protons (p) + number of neutrons (northward)
A= p+ n
Therefore, n= A-p = 13-7 = 6.
Solution using log tabular array-
n= A-p
Applying logten on both sides
log10 n= log (A-Z)
logten northward= logx (13-7) =logten 6=0.7782
Applying antilog,
due north =Antilog (0.7782)
6.001 = half dozen
The number of neutrons in the 13N Isotope is 6, and the number of protons is 7.
14 N Isotope
Mass number =14 (Given)
mass number (A) = number of protons (p) + number of neutrons (n)
A= p+ due north
Therefore, n= A-p = 14-vii = vii.
Solution using log table-
n= A-p
Applying log10 on both sides
logten north= log (A-Z)
log10 northward= logx (xiv-7) = logx 7 =0.8451
Applying antilog,
n =Antilog (0.8451) = vii
The number of neutrons in the 14Due north Isotope is 7, and the number of protons is 7.
15 Due north Isotope
Mass number =fifteen (Given)
mass number (A) = number of protons (p) + number of neutrons (due north)
A= p+ n
Therefore, due north= A-p = fifteen - 7 = 8.
Solution using log tabular array-
northward= A-p
Applying logx on both sides
log10 northward= log (A-Z)
log10 n= log10 (14-7) = logx 8 =0.9031
Applying antilog,
n =Antilog (0.9031) = 8
The number of neutrons in the 15N Isotope is 8, and the number of protons is 7.
16 N Isotope
Mass number =16 (Given)
mass number (A) = number of protons (p) + number of neutrons (n)
A= p+ n
Therefore, due north= A-p = 16 - 7 = nine.
Solution using log table-
n= A-p
Applying log10 on both sides
logx n= log (A-Z)
logten n= log10 (16-7) = log10 9 =0.9542
Applying antilog,
n =Antilog (0.9542) = ix
The number of neutrons in the 16N Isotope is ix, and the number of protons is 7.
17 Due north Isotope
Mass number =17 (Given)
mass number (A) = number of protons (p) + number of neutrons (n)
A= p+ n
Therefore, n= A-p = 17 - seven = 10.
Solution using log tabular array-
n= A-p
Applying log10 on both sides
logten n= log (A-Z)
log10 n= log10 (17-seven) = logx ten =1.000
Applying antilog,
n =Antilog (1.000) = ten
The number of neutrons in the 17Due north Isotope is 10, and the number of protons is vii.
How Many Protons In Nitrogen,
Source: https://www.curlyarrows.com/calculate-number-protons-and-neutrons-nitrogen-isotopes-13n-14n-15n-16n-and-17n
Posted by: turnergother.blogspot.com


0 Response to "How Many Protons In Nitrogen"
Post a Comment