Boiling Point Of Sodium Chloride
| Sodium chloride | |
|---|---|
  | |
| General | |
| Systematic name | Sodium chloride |
| Other names | Common salt, halite, tabular array salt |
| Molecular formula | NaCl |
| Molar mass | 58.442 g/mol |
| Appearance | white and crystalline |
| CAS number | [7647-14-v] |
| Properties | |
| Density and phase | 2.16 m/cm³, solid |
| Solubility in water | 35.9 g/100 ml (25 °C) |
| Melting bespeak | 801 °C (1074 Grand) |
| Boiling point | 1465 °C (1738 K) |
| Structure | |
| Coordination geometry | Octahedral |
| Crystal structure | Confront centered cubic |
| Hazards | |
| MSDS | External MSDS |
| Main hazards | Irritant and may sting |
| NFPA 704 | 0 0 0 |
| Flash point | Non-flammable |
| R/Southward statement | R: none S: none |
| RTECS number | VZ4725000 |
| Supplementary data | |
| Structure and properties | n, εr, etc. |
| Thermodynamic information | Stage behavior Solid, liquid, gas |
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Other anions | NaF, NaBr, NaI |
| Other cations | LiCl, KCl, RbCl, CsCl, MgClii, CaClii |
| Related salts | Sodium acetate |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Sodium chloride, too known every bit common salt or table table salt, is a chemical compound with the formula NaCl. Its mineral form is called halite. It is highly soluble in water and is the salt most responsible for the salinity of the ocean and of the extracellular fluid of many multicellular organisms.
As the master ingredient in edible salt, it has long been used as a food seasoning and preservative. In its latter capacity, it reduced human dependence on the seasonal availability of food and allowed travel over long distances. In this mode, it served every bit a foundation for the spread of civilization.
Contents
- 1 History
- 1.i Religious references
- 2 Occurrence
- 3 Product
- iii.ane Solar evaporation of seawater
- 3.2 Open pan product from brine
- 3.iii Closed pan production under vacuum
- 3.4 Salt mines
- 4 Characteristics
- 4.ane Crystal structure
- 4.2 Solubility
- 5 Uses
- 5.1 Synthetic uses
- 5.ii Flavor enhancer
- 5.three Biological uses
- 6 Road table salt
- 6.1 De-icing
- six.two Additives
- 6.3 Common chemicals
- 7 See also
- 8 Notes
- 9 References
- 10 External links
- xi Credits
It is currently available quite inexpensively and in big quantities. Historically, however, it was hard to obtain and was a highly valued trade item. Until the 1900s, it was one of the prime movers of national economies and wars. Information technology was controlled by governments and taxed equally far back as the twentieth century B.C.E. in China.

Mounds of table salt at Salar de Uyuni, Bolivia
History
Salt'southward preservative ability was a foundation of civilization. It eliminated dependency on the seasonal availability of nutrient and allowed travel over long distances. By the Eye Ages, caravans consisting of as many equally forty grand camels traversed four hundred miles of the Sahara bearing common salt, sometimes trading it for slaves.
During his protests in India, Gandhi led the famous salt march to claiming the British-imposed monopoly on salt.
Religious references
At that place are 35 references (verses) to salt in the Bible (King James Version), the almost familiar probably beingness the story of Lot's wife, who was turned into a pillar of salt when she disobeyed the angels and looked dorsum at the wicked city of Sodom (Genesis 19:26). In the Sermon on the Mount, Jesus also referred to his followers as the "salt of the globe." The campaigner Paul also encouraged Christians to "permit your conversation be always full of grace, seasoned with salt" (Colossians 4:vi) and so that when others inquire well-nigh their beliefs, the Christian'southward respond generates a 'thirst' to know more nearly Christ.
In the native Japanese religion Shinto, salt is used for ritual purification of locations and people, such equally in Sumo Wrestling.
Occurrence
Historically, there have been two main sources for mutual salt: sea h2o and rock salt. Stone common salt occurs in vast beds of sedimentary evaporite minerals that result from the drying up of enclosed lakes, playas, and seas. Table salt beds may be up to 350 meters (meters) thick and underlie wide areas.
In the United States and Canada extensive underground beds extend from the Appalachian bowl of western New York through parts of Ontario and under much of the Michigan basin. Other deposits are in Ohio, Kansas, New United mexican states, Nova Scotia, and Saskatchewan. In the United kingdom of great britain and northern ireland hush-hush beds are found in Cheshire and around Droitwich.
Product

Modern rock salt mine near Mount Morris, New York

Salt is currently produced in one of 2 principal ways:
- The evaporation of seawater or brine (salt water) from other sources, such as brine wells and common salt lakes;[ane]
- The mining of stone salt, chosen halite. This includes solution mining, in which water is used to deliquesce the salt and the brine that reaches the surface is evaporated to recover the salt.
Solar evaporation of seawater
In the correct climate (one for which the ratio of evaporation to rainfall is suitably high) it is possible to use solar evaporation of sea water to produce salt. Alkali is evaporated in a linked set of ponds until the solution is sufficiently concentrated by the final pond that the salt crystallizes on the pond's floor.
Open pan production from alkali
One of the traditional methods of salt product in more than temperate climates is using open up pans.[two] In an open up pan salt works, alkali is heated in large, shallow open up pans. Primeval examples date back to prehistoric times and the pans were fabricated of ceramics known every bit briquetage, or lead. Later examples were made from iron. This change coincided with a change from forest to coal for the purpose of heating the alkali.[three] Brine would be pumped into the pans, and full-bodied by the heat of the fire burning underneath. As crystals of salt formed these would exist raked out and more brine added.
Closed pan production under vacuum
The open pan salt works has effectively been replaced with a closed pan organization where the brine solution is evaporated under a fractional vacuum.[4]
Salt mines
In the second half of the nineteenth century, it became possible to mine salt, which is less expensive than evaporating seawater or extracting common salt from brine. Consequently, the price of salt became more reasonable. However, extraction of common salt from brine is even so heavily used: for example vacuum salt produced by British Salt in Middlewich has 57 percent of the UK market place [5] for table salt used in cooking.
Characteristics
Crystal construction
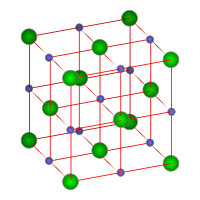
The crystal structure of sodium chloride. Each atom has six nearest neighbors, with octahedral geometry.
Sodium chloride forms crystals with cubic symmetry. In these, the larger chloride ions, shown in the diagram equally green spheres, are arranged in a cubic close-packing, while the smaller sodium ions, shown as blueish spheres, fill the octahedral gaps between them. The ions are held together with ionic bonds.
Each type of ion is surrounded by six ions of the other kind. This aforementioned basic structure is institute in many other minerals and is known every bit the halite structure. This arrangement is known every bit cubic shut packed (ccp) crystal organization.
Solubility
| Solubility of NaCl in various solvents (grams NaCl per 100 grams of solvent at 25 °C) | |
|---|---|
| H2O | 36 |
| Liquid ammonia | 3.02 |
| Methanol | ane.4 |
| Formic acid | 5.2 |
| Sulfolane | 0.005 |
| Acetonitrile | 0.0003 |
| Acetone | 0.000042 |
| Formamide | 9.4 |
| Dimethylformamide | 0.04 |
| Reference: J. Burgess, Metal Ions in Solution (Ellis Horwood, New York, 1978), ISBN 0853120277 | |
Uses
While most people are familiar with the many uses of salt in cooking, they might be unaware that table salt is used in a plethora of applications, from manufacturing pulp and newspaper to setting dyes in textiles and fabric, to producing soaps and detergents. In well-nigh of Canada and the northern U.s., large quantities of rock table salt are used to assistance articulate highways of ice during winter, although "route table salt" loses its melting ability at temperatures beneath -xv °C to -20 °C (5 °F to -4 °F).
Synthetic uses
Salt is also the raw material used to produce chlorine which itself is required for the production of many modern materials including PVC and pesticides.
Industrially, elemental chlorine is usually produced by the electrolysis of sodium chloride dissolved in water. Forth with chlorine, this chloralkali process yields hydrogen gas and sodium hydroxide, according to the chemic equation:
- 2NaCl + 2HiiO → Cl2 + Hii + 2NaOH
Sodium metal is produced commercially through the electrolysis of liquid sodium chloride. This is done in an apparatus called a Downs jail cell, in which sodium chloride is mixed with calcium chloride to lower the melting point below 700 °C. As calcium is more electropositive than sodium, no calcium will be formed at the cathode. This method is less expensive than the earlier method of electrolyzing sodium hydroxide.
Flavour enhancer
Salt is unremarkably used as a flavor enhancer for food and has been identified equally one of the basic tastes. Unfortunately, it is frequently ingested well in excess of the required intake. This leads to elevated levels of blood pressure level (hypertension) in some, which in turn is associated with increased risks of centre attack and stroke. Consuming salt in excess tin besides dehydrate the human body.
Biological uses
Many microorganisms cannot live in an overly salty environment: h2o is drawn out of their cells by osmosis. For this reason salt is used to preserve some foods, such as smoked bacon or fish, and can also be used to disassemble leeches that have attached themselves to feed. It has also been used to disinfect wounds (although it causes a slap-up deal of pain). In medieval times, salt would be rubbed into household surfaces as a cleansing amanuensis.
Road salt
De-icing
While salt was one time a deficient commodity in history, industrialized product has at present made common salt plentiful. Near 51 per centum of the worldwide output of common salt is at present used to de-ice roads in freezing atmospheric condition atmospheric condition. The salt may exist put in grit bins and spread by winter service vehicles. This approach works considering salt and water form an eutectic mixture. Nether controlled lab atmospheric condition, a solution of sodium chloride in water can reduce the freezing temperature of water to -21 °C (-6 °F). In practice, however, sodium chloride can melt ice down to only virtually -9 °C (15 °F).
Additives
The salt sold for consumption today is usually not pure sodium chloride. In 1911, magnesium carbonate was first added to salt to make it period more freely. In 1924, trace amounts of iodine—in the form of sodium iodide, potassium iodide or potassium iodate—were kickoff added, creating iodized common salt to reduce the incidence of unproblematic goiter.
Salt for de-icing in the UK typically contains sodium hexacyanoferrate (II) at less than 100 parts per meg as an anti-caking agent. In recent years this additive has also been used in table common salt.
Common chemicals
Chemicals used in de-icing salts are mostly found to exist sodium chloride (NaCl) or calcium chloride (CaCltwo). Both are like and are effective in de-icing roads. When these chemicals are produced, they are mined/fabricated, crushed to fine granules, then treated with an anti-caking amanuensis. Calculation salt lowers the freezing bespeak of the water, which allows the liquid to be stable at lower temperatures and allows the ice to cook.
Alternative de-icing chemicals have also been used. Chemicals such as calcium magnesium acetate are being produced. These chemicals have few of the negative chemical effects on the surround unremarkably associated with NaCl and CaCl2.
See too
- Black salt
- Edible table salt
- Halite
- Salinity
- Soap
Notes
- ↑ Solar Salt production. Salt Institute. Retrieved June 22, 2007.
- ↑ Towards an understanding of open pan salt making. Lion Salt Works History & Heritage. Retrieved June 22, 2007.
- ↑ Early Salt Making. Lion Common salt Works History & Heritage. Retrieved June 22, 2007.
- ↑ Vacuum Pan Salt Refining. Salt Institute. Retrieved June 22, 2007.
- ↑ The Competition Commission. Factors affecting rivalry in the relevant marketplace prior to the merger. British Salt Limited and New Cheshire Table salt Works Express: A study on the acquisition past British Salt Limited of New Cheshire Salt Works Limited. Retrieved June 22, 2007.
References
ISBN links support NWE through referral fees
- Kurlansky, Mark. 2003. Salt: A World History. New York: Penguin. ISBN 0142001619
- Multhauf, Robert P. 1996. Neptune's Gift: A History of Common Salt. Johns Hopkins Studies in the History of Technology. Baltimore, Doctor: Johns Hopkins University Press. ISBN 0801854695
- Mineral Gallery. 2006. The Mineral Halite. Amethyst Galleries. Retrieved May 9, 2007.
External links
All links retrieved November sixteen, 2019.
- MrBloch Salt Archive
- Table salt: Statistics and Information. U.s. Geological Survey.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Artistic Commons CC-past-sa iii.0 License (CC-past-sa), which may exist used and disseminated with proper attribution. Credit is due nether the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click hither for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
- Sodium_chloride history
- History_of_salt history
The history of this article since information technology was imported to New Earth Encyclopedia:
- History of "Sodium chloride"
Annotation: Some restrictions may apply to utilize of individual images which are separately licensed.
Boiling Point Of Sodium Chloride,
Source: https://www.newworldencyclopedia.org/entry/Sodium_chloride
Posted by: turnergother.blogspot.com



0 Response to "Boiling Point Of Sodium Chloride"
Post a Comment